| The ebook FEEE - Fundamentals of Electrical Engineering and Electronics is based on material originally written by T.R. Kuphaldt and various co-authors. For more information please read the copyright pages. |

|

Home  Semiconductors Semiconductors  Solid-State Device Theory Solid-State Device Theory  Quantum Physics Quantum Physics  Angular Momentum Quantum Number Angular Momentum Quantum Number |
||






|
||
|
Angular Momentum Quantum Number
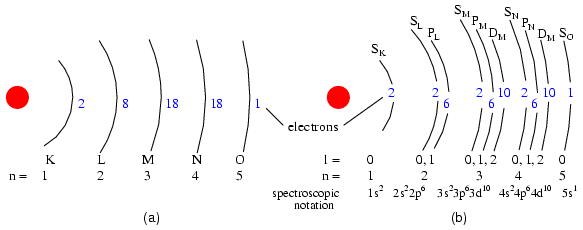
Orbitals: (s) Three fold symmetry. (p) Shown: sx, one of three possible orientations (sx, sy, sz ), about their respective axes. (d) Shown: dx2-y x2 similar to dxz, dyz, dxz. Shown: dz2. Possible d-orbital orientations: five. Valid angular momentum quantum numbers are positive integers like principal quantum numbers, but also include zero. These quantum numbers for electrons are symbolized by the letter l. The number of subshells in a shell is equal to the shell's principal quantum number. Thus, the first shell (n=1) has one subshell, numbered 0; the second shell (n=2) has two subshells, numbered 0 and 1; the third shell (n=3) has three subshells, numbered 0, 1, and 2. An older convention for subshell description used letters rather than numbers. In this notation, the first subshell (l=0) was designated s, the second subshell (l=1) designated p, the third subshell (l=2) designated d, and the fourth subshell (l=3) designated f. The letters come from the words sharp, principal (not to be confused with the principal quantum number, n), diffuse, and fundamental. You will still see this notational convention in many periodic tables, used to designate the electron configuration of the atoms' outermost, or valence, shells. (Figure below) (a) Bohr representation of Silver atom, (b) Subshell representation of Ag with division of shells into subshells (angular quantum number l). This diagram implies nothing about the actual position of electrons, but represents energy levels.
|
||
Home  Semiconductors Semiconductors  Solid-State Device Theory Solid-State Device Theory  Quantum Physics Quantum Physics  Angular Momentum Quantum Number Angular Momentum Quantum Number |
||
Last Update: 2010-11-19