| The ebook FEEE - Fundamentals of Electrical Engineering and Electronics is based on material originally written by T.R. Kuphaldt and various co-authors. For more information please read the copyright pages. |

|

Home  Semiconductors Semiconductors  Solid-State Device Theory Solid-State Device Theory  Electrons and Holes Electrons and Holes  Intrinsic Semiconductor Intrinsic Semiconductor |
|






|
|
|
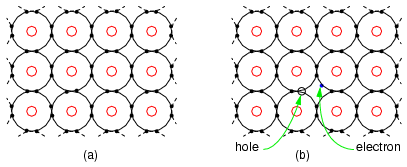
Intrinsic SemiconductorPure semiconductors are relatively good insulators as compared with metals, though not nearly as good as a true insulator like glass. To be usefull in semiconductor applications, the intrinsic semiconductor, pure undoped semiconductor must have no more than one impurity atom in 10 billion semiconductor atoms. This is analogous to a grain of salt impurity in a railroad boxcar of sugar. Impure, or dirty semiconductors are considerably more conductive, though not as good as metals. Why might this be? To answer that question, we must look at the electron structure of such materials in Figure below. Figure below (a) shows four electrons in the valence shell of a semiconductor forming covalent bonds to four other atoms. This is a flattened, easier to draw, version of Figure above. All electrons of an atom are tied up in four covalent bonds, pairs of shared electrons. Electrons are not free to move about the crystal lattice. Thus, intrinsic, pure, semiconductors are relatively good insulators as compared to metals. (a) Intrinsic semiconductor is an insulator having a complete electron shell. (b) However, thermal energy can create few electron hole pairs resulting in weak conduction. Thermal energy may occasionally free an electron from the crystal lattice as in Figure above (b). This electron is free for conduction about the crystal lattice. When the electron was freed, it left an empty spot with a positive charge in the crystal lattice known as a hole. This hole is not fixed to the lattice; but, is free to move about. The free electron and hole both contribute to conduction about the crystal lattice. That is, the electron is free until it falls into a hole. This is called recombination. If an external electric field is applied to the semiconductor, the electrons and holes will conduct in opposite directions. Increasing temperature will increase the number of electrons and holes, decreasing the resistance. This is opposite of metals, where resistance increases with temperature by increasing the collisions of electrons with the crystal lattice. The number of electrons and holes in an intrinsic semiconductor are equal. However, both carriers do not necessarily move with the same velocity with the application of an external field. Another way of stating this is that the mobility is not the same for electrons and holes. Pure semiconductors, by themselves, are not particularly usefully. Though, semiconductors must be refined to a high level of purity as a starting point prior the addition of specific impurities.
|
|
Home  Semiconductors Semiconductors  Solid-State Device Theory Solid-State Device Theory  Electrons and Holes Electrons and Holes  Intrinsic Semiconductor Intrinsic Semiconductor |
|
Last Update: 2010-11-19