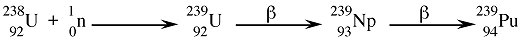| General Chemistry is a free introductory textbook on chemistry. See the editorial for more information.... |

|

Home  Nuclear Chemistry Nuclear Chemistry  Nuclear Energy Nuclear Energy |
|||||
| See also: Nuclear Fission, Nuclear Fusion | |||||






|
|||||
Nuclear EnergyAuthor: Andrew R. Barron
Despite the horrific power of the fission bombs it was realized that if the fission reaction was controlled a vast amount of energy could be obtained from relatively small amounts of material. In a nuclear power plant the chain reaction is controlled through the absorption of the neutrons by control rods made of chemical elements capable of absorbing neutrons without undergoing fission themselves. These elements include silver, indium, cadmium, boron, cobalt, hafnium, or their alloys and compounds. The nuclear material is present in fuel rods between which the control rods moderate the flux of neutrons. The heat generated from the fission process is not used directly to generate electricity, but via a suitable heat exchanger it heats water to steam that in turn drives a turbine-generator. Despite the large amount of energy potentially generated by nuclear fission there are several potential problems. Real and imagined dangerGiven the similarity of nuclear chemical reactions in a fission reactor and an atom bomb there is a misguided perception that a nuclear power plant could blow up in an atom bomb type explosion. The low amount of uranium-235 used in the fuel rods is below the critical mass to allow for a nuclear explosion. In comparison, a real risk does exist in which a system or component failure causes the reactor core to cease being properly controlled and cooled to the extent that the sealed nuclear fuel assemblies (which contain the uranium or plutonium and highly radioactive fission products) begin to overheat and melt. The resulting meltdown is considered very serious because of the possibility that the reactor containment will be defeated, thus releasing the core's highly radioactive and toxic elements into the atmosphere and environment. Although several potential meltdowns have been reported, there have only been two actual significant meltdown events. The first was in 1979 at the Three Mile Island Nuclear Generating Station in Dauphin County, Pennsylvania (USA). It was the most significant accident in the history of the American commercial nuclear power generating industry, resulting in the release of up to 13 million curies of radioactive gases. A failure in the non-nuclear secondary system, followed by a stuck-open relief valve in the primary system, allowed large amounts of reactor coolant to escape. This resulted in the core reaching 2000 °C: very close to a meltdown, however, the reactor was brought under control. In contrast when the chain reaction grew out of control Chernobyl Nuclear Power Plant in Ukraine, then part of the Soviet Union, a steam explosion resulted. This was followed by a second chemical explosion from the ignition of generated hydrogen mixing with air, which tore the top from the reactor and its building, and exposed the reactor core. This sent large amounts of radioactive particulate and gaseous debris into the air. The open core also allowed air to contact the super-hot core containing 1,700 tonnes of combustible graphite moderator. The burning graphite moderator increased the emission of radioactive particles, carried by the smoke.(1)
Available supply of fissionable materialsUranium is present in the Earth's crust at a concentration of 2 - 4 parts per million, and as such it is more abundant than antimony, tin, cadmium, mercury, or silver, and it is about as abundant as arsenic or molybdenum. However, in nature uranium atoms exist as uranium-238 (99.284%), uranium-235 (0.711%), and a very small amount of uranium-234 (0.0058%). Thus, in order to obtain sufficient uranium-235 a large quantity of uranium ore must be mined, and while nuclear generated electricity may be considered relatively environmentally friendly as compared to the combustion of hydrocarbons, the mining process has a significant environmental cost. It is for this reason that so-called fast-breeder reactors were developed. By wrapping a plutonium-239 core with uranium-238 the neutrons generated could be used to covert abundant uranium-238 to the fissionable plutonium-239:
Disposal of wasteWhile it has been estimated that a coal power plant releases 100 times as much radiation as a nuclear power plant of the same wattage, and in 1982 US coal burning released 155 times as much radioactivity into the atmosphere as the Three Mile Island accident, the high level radioactive waste resulting from nuclear power stations does represent a major concern. In particular the long storage times needed (on the order of 10,000 years) posses significant issues. CostIrrespective of the reactor designs, nuclear is a more expensive method of electricity production than coal or natural gas. However, with the high environmental (CO2) impact and pollution concerns of coal burning the balance is swinging to nuclear energy.
|
|||||
Home  Nuclear Chemistry Nuclear Chemistry  Nuclear Energy Nuclear Energy |
|||||
Last Update: 2011-02-20